Healthcare & Pharmaceutical Solutions: From Talent Acquisition to Blockchain Supply Chains
Healthcare Technology Challenges
Healthcare and pharmaceutical organizations operate under unique constraints that make technology implementation particularly challenging:
- Regulatory Compliance: HIPAA, JCAHO, FDA regulations with severe penalties for violations
- Data Sensitivity: Protected health information (PHI), patient privacy, clinical data security
- Legacy Systems: Decades-old systems that cannot be easily replaced but must integrate with modern platforms
- High Stakes: Technology failures can impact patient safety, drug efficacy tracking, and public health
- Complexity: Multi-stakeholder coordination (providers, payers, manufacturers, distributors, regulators)
Since 2009, we’ve been building technology solutions for this demanding sector, focusing on reliability, compliance, and security as foundational requirements—not afterthoughts.
Healthcare Talent Acquisition: The Nurse Portal
The Credentialing Challenge
Healthcare facilities face a critical shortage of clinical staff combined with complex credentialing requirements. Hiring a registered nurse isn’t just posting a job and conducting interviews—it requires:
License Verification:
- State nursing board verification (RN, LPN, APRN licenses)
- Multi-state license compact (NLC) validation
- Specialty certifications (CCRN, ACLS, PALS, etc.)
- Expiration tracking and renewal alerts
Background Screening:
- Criminal background checks (state and federal)
- Employment history verification
- Professional reference checks
- OIG exclusion list screening (Medicare/Medicaid fraud)
- Abuse registry checks
Credentialing Documentation:
- Educational transcripts
- Continuing education credits
- Immunization records
- TB test results
- Physical exam documentation
Compliance Requirements:
- JCAHO standards for credentialing
- State-specific regulations
- Hospital-specific policies
- Audit trail for regulatory inspections
The Solution: Integrated Talent Acquisition Platform
We built a comprehensive platform connecting talent acquisition, credentialing, and onboarding workflows for a major healthcare organization.
Technology Stack:
- Application Server: Glassfish 4 (Java EE 7)
- Workflow: Camunda BPMN for credentialing process orchestration
- Frontend: PrimeFaces (JSF components) for rich user interface
- Database: Oracle 12c with encrypted PHI storage
- Integration: REST/SOAP web services for license boards, background check providers, HRIS
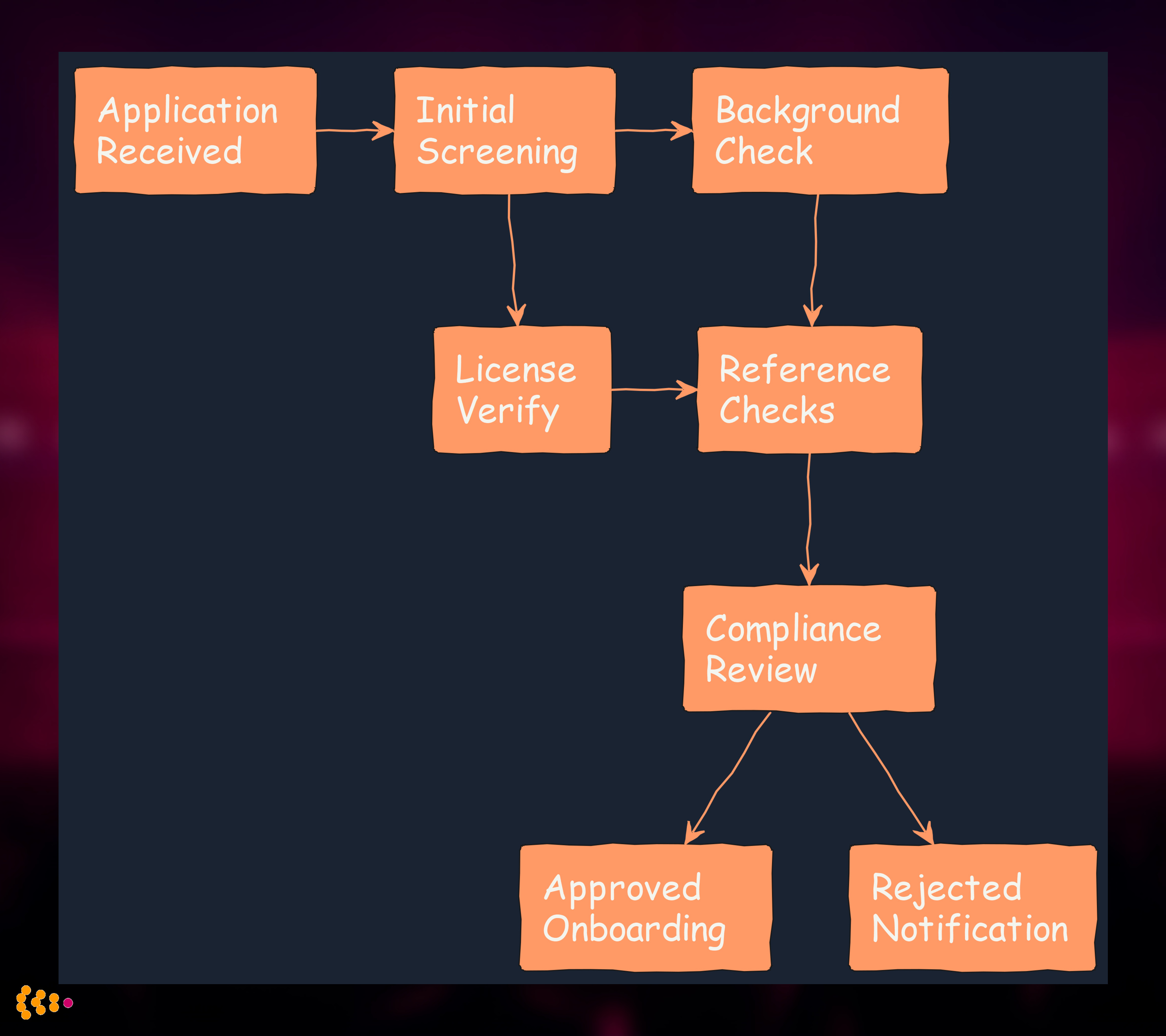
Credentialing Workflow (Camunda BPMN):
Key Features:
Automated License Verification: Direct API integration with state nursing boards—system queries license status, expiration date, and disciplinary actions automatically.
Intelligent Task Routing: Camunda workflow routes tasks based on role type (RN vs. physician vs. therapist), urgency, and compliance specialist availability.
Compliance Dashboard: Real-time view of credentialing pipeline showing bottlenecks, pending verifications, and approaching license expirations.
Audit Trail: Every action logged with timestamp, user, and reason—essential for JCAHO inspections and legal discovery.
Results
Time Reduction: Credentialing cycle reduced from 45 days to 18 days average (60% improvement).
Compliance: 100% pass rate on JCAHO audits over 3-year period—complete audit trails and documentation.
Volume: Platform processed 2,000+ candidates annually across 3 hospital systems.
User Satisfaction: 95% of recruiters rated platform as “excellent” or “very good” within 3 months of deployment.
Cost Savings: Eliminated 2 FTE positions in manual data entry and verification coordination.
Pharmaceutical Supply Chain Tracing with Blockchain
The pharmaceutical industry faces a critical challenge: counterfeit drugs entering the supply chain. The WHO estimates that 10% of medications in developing countries are fake, and even developed markets see 1-2% counterfeit penetration. The consequences range from treatment ineffectiveness to patient deaths.
The Drug Supply Chain Security Act (DSCSA)
U.S. FDA’s DSCSA mandates full track-and-trace capability for prescription drugs by 2023 (later extended). Requirements include:
- Serialization: Unique identifier for each saleable unit
- Verification: Ability to verify legitimacy at each supply chain step
- Traceability: Complete custody chain from manufacturer to patient
- Interoperability: Data sharing across manufacturers, wholesalers, dispensers
Traditional approaches (centralized databases, EDI) struggle with:
- Trust: Who controls the database? How to prevent tampering?
- Scalability: Billions of transactions annually across thousands of entities
- Interoperability: Competing systems, proprietary formats, data silos
Blockchain Solution: Hyperledger Fabric
We led development of a blockchain-based supply chain tracing platform using Hyperledger Fabric—a permissioned blockchain framework designed for enterprise use.
Why Blockchain:
- Immutability: Once recorded, custody transfers cannot be altered (tamper-proof)
- Decentralization: No single entity controls the ledger (trust distribution)
- Transparency: All participants can verify custody chain (while maintaining privacy)
- Smart Contracts: Automated verification logic (chaincode in Hyperledger)
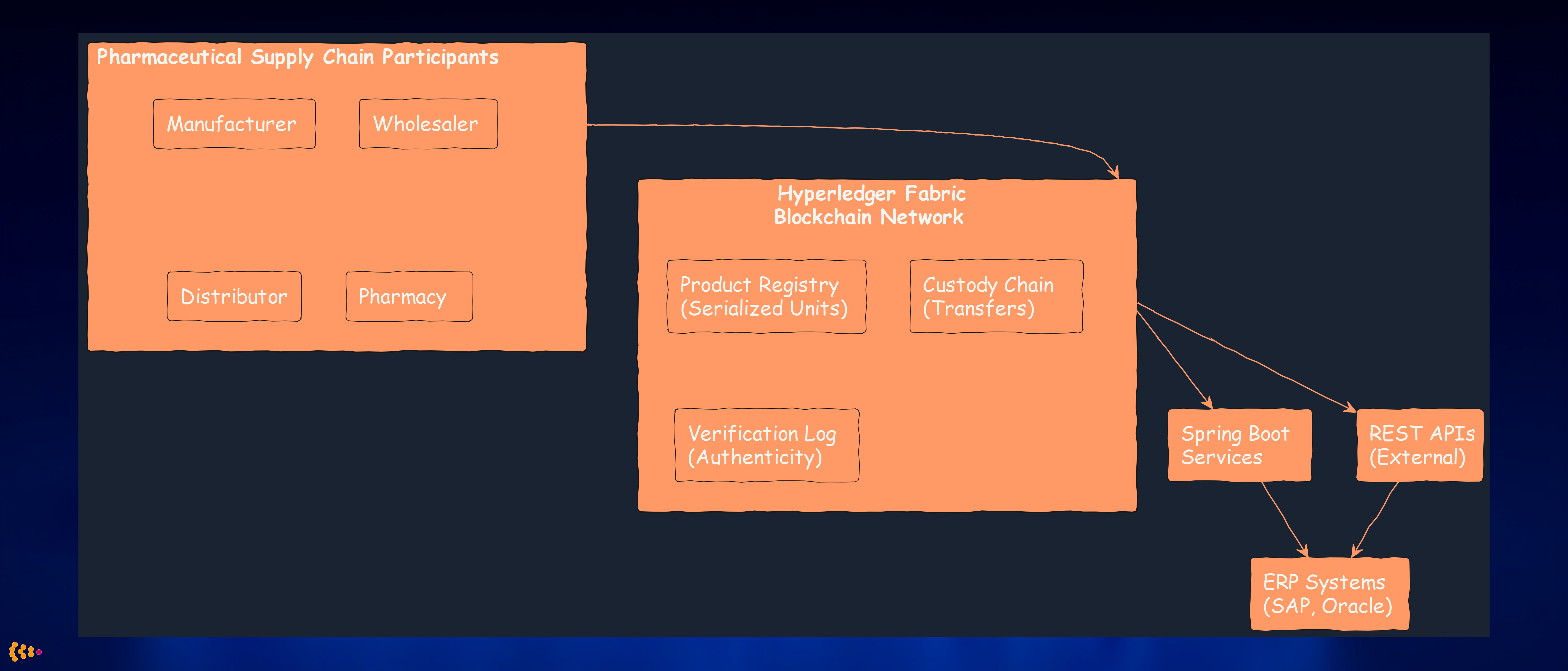
Architecture Highlights:
Permissioned Network: Only authorized entities (manufacturers, wholesalers, etc.) can join network—unlike public blockchains.
Channels: Private sub-networks for confidential data (e.g., pricing negotiated between manufacturer and wholesaler stays private).
Smart Contracts (Chaincode): Business logic enforcing custody rules—e.g., pharmacy cannot mark product “verified” unless previous custody was wholesaler.
Off-Chain Storage: Bulk data (product descriptions, images) stored in PostgreSQL; blockchain stores hashes and custody events for immutability.
Technology Stack:
- Blockchain: Hyperledger Fabric 1.4+
- Application Layer: Spring Boot microservices
- Database: PostgreSQL (off-chain data), Fabric ledger (on-chain custody)
- Integration: Kafka for event streaming, REST APIs for ERP connectivity
- DevOps: Docker, Kubernetes, GitLab CI/CD
Implementation Details
Product Registration (on blockchain):
- Manufacturer registers serialized product (GTIN + serial number + batch + expiration)
- Blockchain creates immutable record with timestamp and digital signature
- Hash of product data stored on-chain; full data in PostgreSQL
Custody Transfer:
- Manufacturer ships to wholesaler → blockchain records transfer with GPS coordinates, timestamp
- Wholesaler receives and verifies → scans barcode, confirms receipt on blockchain
- Each transfer creates new block with custody chain reference
Verification:
- Pharmacy scans product barcode → queries blockchain for custody history
- System shows: manufacturer → wholesaler → distributor → pharmacy (complete chain)
- Any gap or anomaly (e.g., unexpected custody transfer) triggers alert
Results
Compliance: Full FDA DSCSA compliance—100% serialization and traceability.
Security: Zero data tampering incidents over 2-year production period (blockchain immutability).
Availability: 99.9% system uptime with Kubernetes orchestration and Fabric high-availability configuration.
Performance: 50,000+ supply chain events processed daily with sub-second blockchain query response.
Counterfeit Detection: 3 counterfeit shipments detected and intercepted before reaching pharmacies (custody chain anomalies flagged by system).
Regulatory: Passed FDA audit with full audit trail and tamper-proof custody records.
Read Full Blockchain Case Study →
Pharmaceutical Market Research Analytics
Market research in pharma requires sophisticated analysis of clinical trial data, prescription trends, market share, and competitive intelligence.
The Challenge
A pharmaceutical market research firm needed to:
- Analyze large datasets (millions of prescriptions, clinical trial results)
- Generate statistical models for market forecasting
- Provide interactive dashboards for client deliverables
- Support multiple data formats (CSV, SPSS, SAS, proprietary databases)
- Ensure data confidentiality (client proprietary data, PHI from prescription databases)
Custom Analytics Platform
We developed a platform combining:
Data Integration: Connectors for various data sources (prescription databases, clinical trial registries, market research panels)
Statistical Analysis: Integration with R and Python for advanced analytics (survival analysis for clinical trials, time series forecasting for prescription trends)
Visualization: Interactive dashboards with drill-down capabilities (Highcharts, D3.js)
Reporting: Automated report generation with customizable templates (Jasper Reports, LaTeX for scientific publications)
Security: Role-based access control, data encryption, audit logging for compliance
Technology Stack:
- Backend: Java/Spring Framework
- Analytics: R (statistical computing), Python (pandas, scikit-learn)
- Database: PostgreSQL with partitioning for large datasets
- Frontend: ExtJS for data-heavy interactive grids
- Reporting: Jasper Reports, LaTeX
Results
Analysis Speed: 75% reduction in time from data ingestion to final analysis (automation of data cleaning and model execution).
Accuracy: Eliminated manual data entry errors through automated validation and transformation pipelines.
Client Satisfaction: Market research firm reported “significant improvement in deliverable quality and turnaround time.”
Scalability: Platform supported analysis of datasets 10x larger than previous manual Excel-based workflows.
Market Research Tool Development
Beyond pharmaceutical-specific analytics, we’ve developed general-purpose market research tools for survey analysis, panel management, and statistical modeling.
Survey Platform
Features:
- Survey design with branching logic and randomization
- Multi-language support for international studies
- Panel management (recruitment, quota sampling, weighting)
- Real-time response tracking and quality control
- Statistical analysis (crosstabs, significance testing, factor analysis)
- Automated report generation with charts and tables
Technology: Java/J2EE, PostgreSQL, JavaScript (survey rendering), R (statistical analysis)
Use Cases:
- Pharmaceutical awareness studies (physician surveys on drug familiarity)
- Patient satisfaction surveys (hospital experience, treatment outcomes)
- Market segmentation research (consumer attitudes toward health products)
Data Quality Tools
Features:
- Response time analysis (detect speeders—respondents rushing through surveys)
- Consistency checks (flag contradictory responses)
- Open-end text analysis (NLP to detect gibberish or off-topic responses)
- Bot detection (identify automated survey responses)
Impact: Improved data quality scores from 85% “clean” responses to 96%+ through automated quality controls.
Common Themes: Robust JEE/Java Platforms
All our healthcare and pharma solutions share a common foundation:
Technology Choices Driven by Requirements:
- Java/JEE: Mature, enterprise-grade, extensive library ecosystem
- Application Servers (Glassfish, Payara): Clustering, transaction management, security out-of-the-box
- Relational Databases (Oracle, PostgreSQL): ACID compliance for critical healthcare data
- Workflow Engines (Camunda BPMN): Complex multi-step processes with audit trails
- Spring Boot: Modern microservices for new components, gradual migration from monoliths
Non-Functional Requirements as First-Class Citizens:
- Security: Encryption at rest and in transit, role-based access, audit logging
- Compliance: HIPAA, JCAHO, FDA regulations baked into system design
- Reliability: 99.9% uptime targets, clustering, automated failover
- Performance: Sub-second response times for user interactions, batch processing for large datasets
- Maintainability: Comprehensive documentation, automated tests, clean architecture
Integration Patterns:
- REST/SOAP Web Services: Integrating with external systems (HRIS, background check providers, ERP)
- Message Queues (JMS, Kafka): Asynchronous processing, event-driven architectures
- Batch Processing: Spring Batch for ETL, report generation, data synchronization
Lessons Learned
1. Regulatory Compliance Cannot Be Retrofitted
Design for compliance from day one. Adding HIPAA compliance to an existing system is exponentially harder than building it in from the start.
Example: Audit logging—every data access must be logged with user, timestamp, purpose. Retrofitting this requires touching every database query and service call.
2. Healthcare Workflows Are Complex—Model Them Explicitly
Don’t hide workflows in code. Use BPMN (Camunda) to model processes visually. This enables:
- Business stakeholder review: Non-technical users can validate workflow logic
- Audit transparency: Regulators can see process flows in inspections
- Flexibility: Changing credentialing requirements doesn’t require code changes, just BPMN updates
3. Legacy Integration Is the Norm, Not the Exception
Healthcare organizations run systems from the 1980s-90s that cannot be replaced. Successful projects embrace integration with legacy systems rather than demanding wholesale replacement.
Example: HL7 interfaces for EMR/EHR systems—ancient protocol, but still universal in healthcare. Build HL7 adapters, don’t fight it.
4. Data Quality Is Life-or-Death in Pharma
Counterfeit drugs kill people. License verification errors delay patient care. Data quality isn’t a nice-to-have—it’s the core requirement.
Approach: Multi-layer validation (client-side, server-side, database constraints), automated verification against authoritative sources (state boards, FDA databases), human review for edge cases.
5. Blockchain Is Powerful But Not a Silver Bullet
Blockchain solves specific problems (immutable audit trail, multi-party trust). It’s not a database replacement or performance optimizer.
When to use blockchain: Multi-party scenarios where no single entity should control the ledger (pharmaceutical supply chain, clinical trial data sharing).
When NOT to use blockchain: Single-organization applications, high-throughput transactional systems, use cases where traditional databases suffice.
The Future of Healthcare Technology
Interoperability: FHIR (Fast Healthcare Interoperability Resources) becoming standard for health data exchange. Our future projects will prioritize FHIR APIs.
AI in Healthcare: Predictive analytics for patient outcomes, diagnostic assistance, drug discovery. Our “AI Second” philosophy applies—robust processes first, AI augmentation second.
Blockchain Beyond Pharma: Clinical trial data provenance, medical credential verification, insurance claims processing.
Telehealth: Post-pandemic acceleration of remote care—integration with telehealth platforms and remote patient monitoring devices.
Cloud Migration: Shifting from on-premise to cloud (Azure, AWS) while maintaining HIPAA compliance and data sovereignty requirements.
Conclusion
Since 2009, we’ve supported healthcare and pharmaceutical organizations with technology that meets their unique demands: reliability, compliance, security, and integration with legacy systems.
From talent acquisition platforms streamlining nurse credentialing to blockchain supply chains preventing counterfeit drugs, our solutions prioritize business outcomes over technology novelty.
Our foundation—robust JEE/Java platforms with proven application servers, workflow engines, and integration frameworks—provides the stability healthcare organizations need while enabling innovation where it matters.
Technologies Used: Java, JEE, Glassfish, Payara, Spring Boot, Camunda BPMN, Hyperledger Fabric, Oracle, PostgreSQL, PrimeFaces, ExtJS, R, Python, Kafka, Docker, Kubernetes, REST/SOAP, HL7, FHIR
Industries: Healthcare (hospitals, health systems, talent acquisition), Pharmaceuticals (supply chain, market research, analytics)
Compliance: HIPAA, JCAHO, FDA DSCSA, GxP
Related Case Studies:
Contact: Building healthcare or pharmaceutical technology solutions? Let’s discuss your compliance and integration needs.